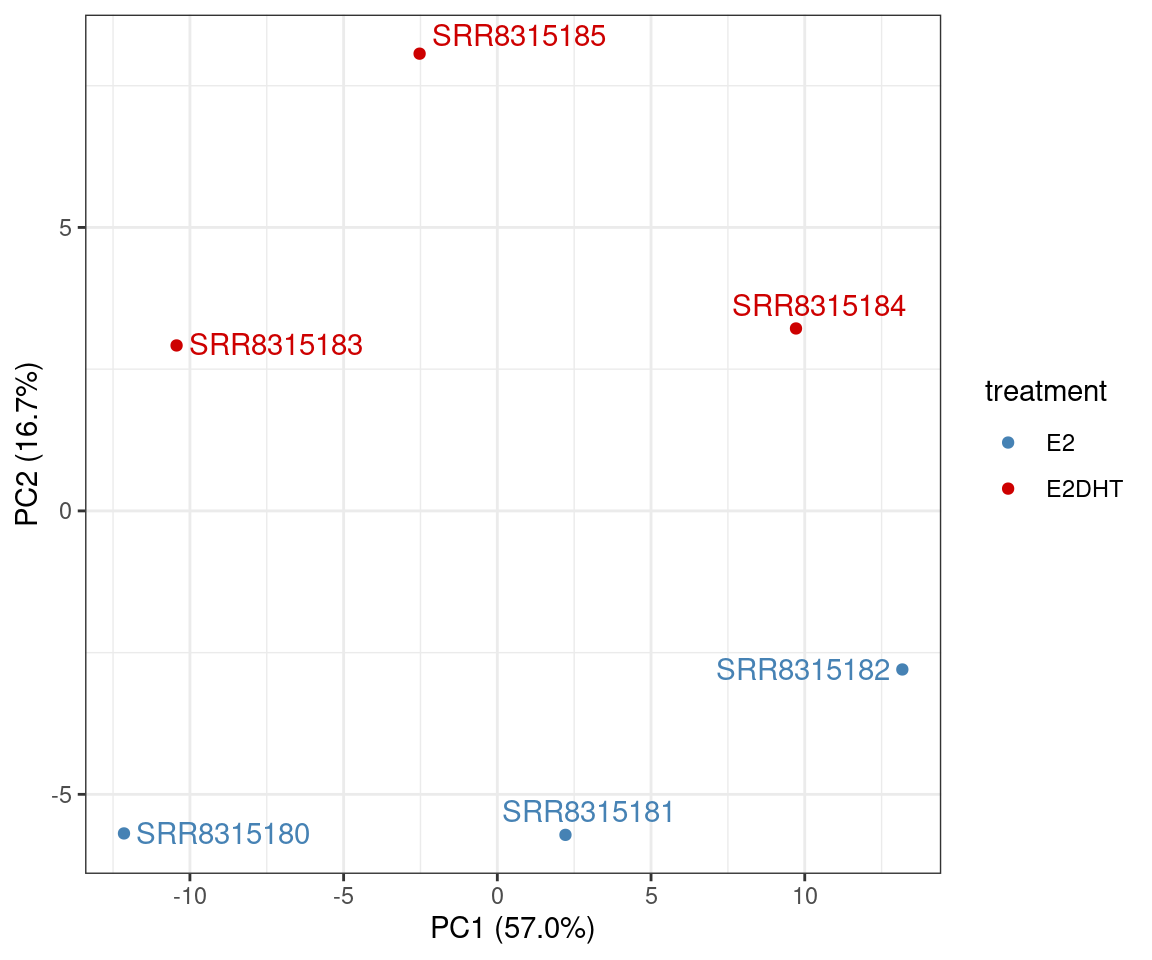
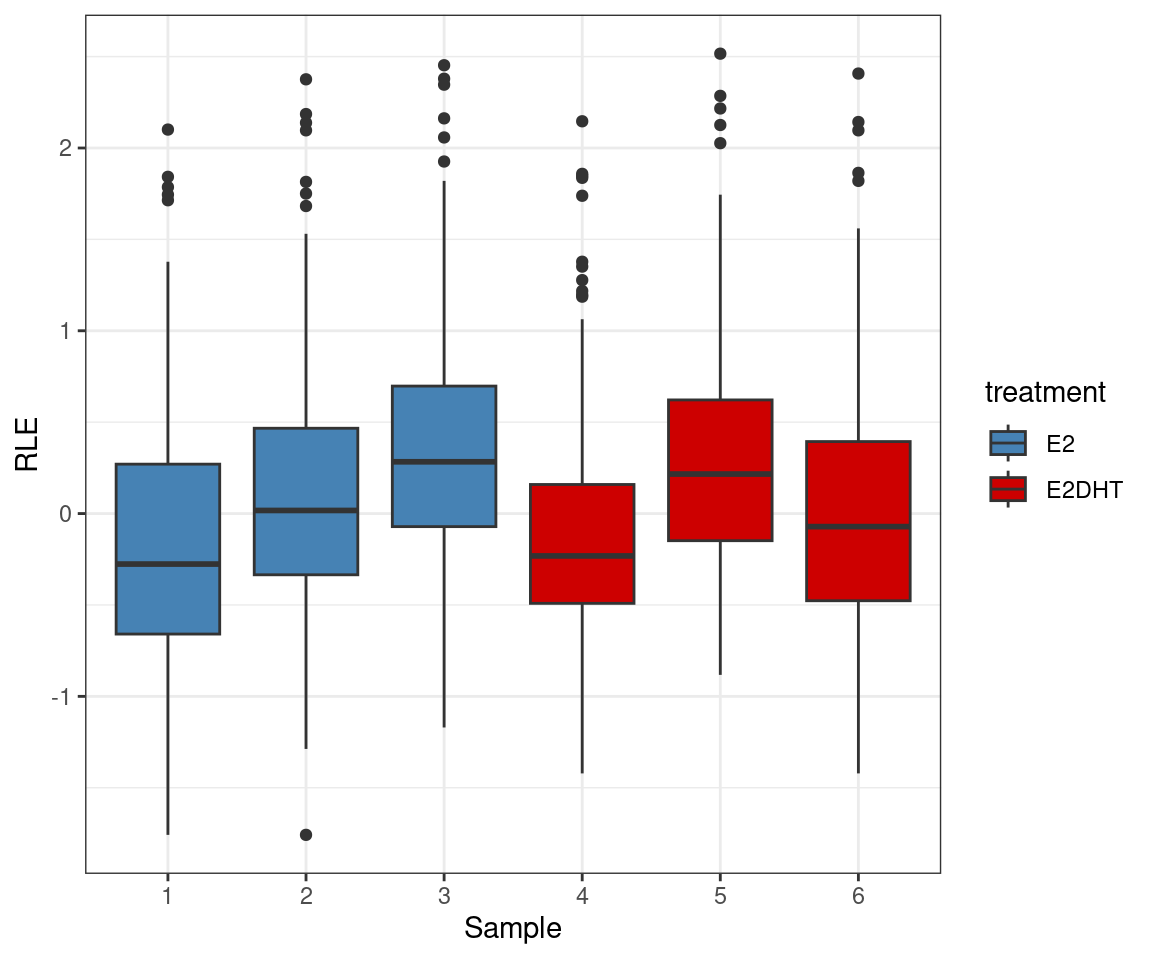
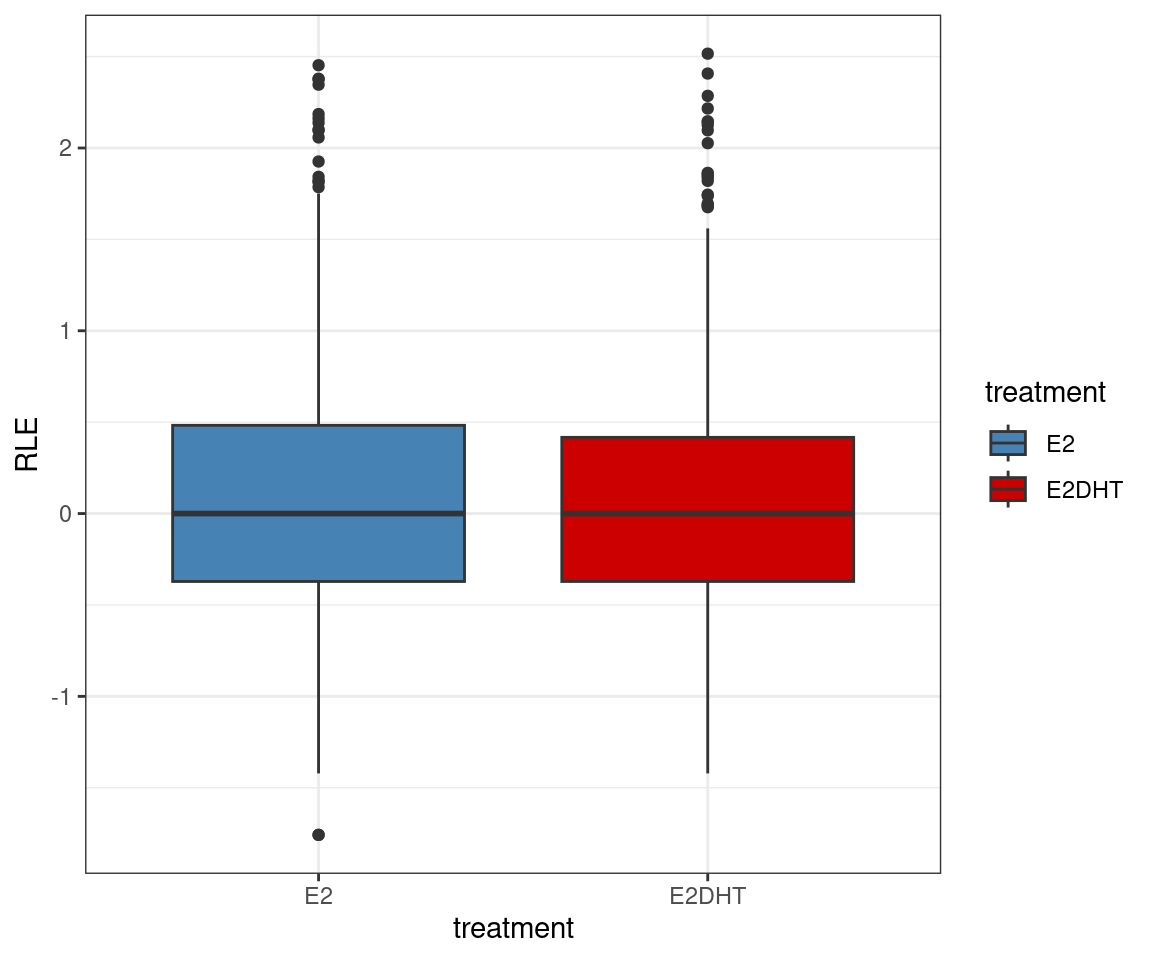
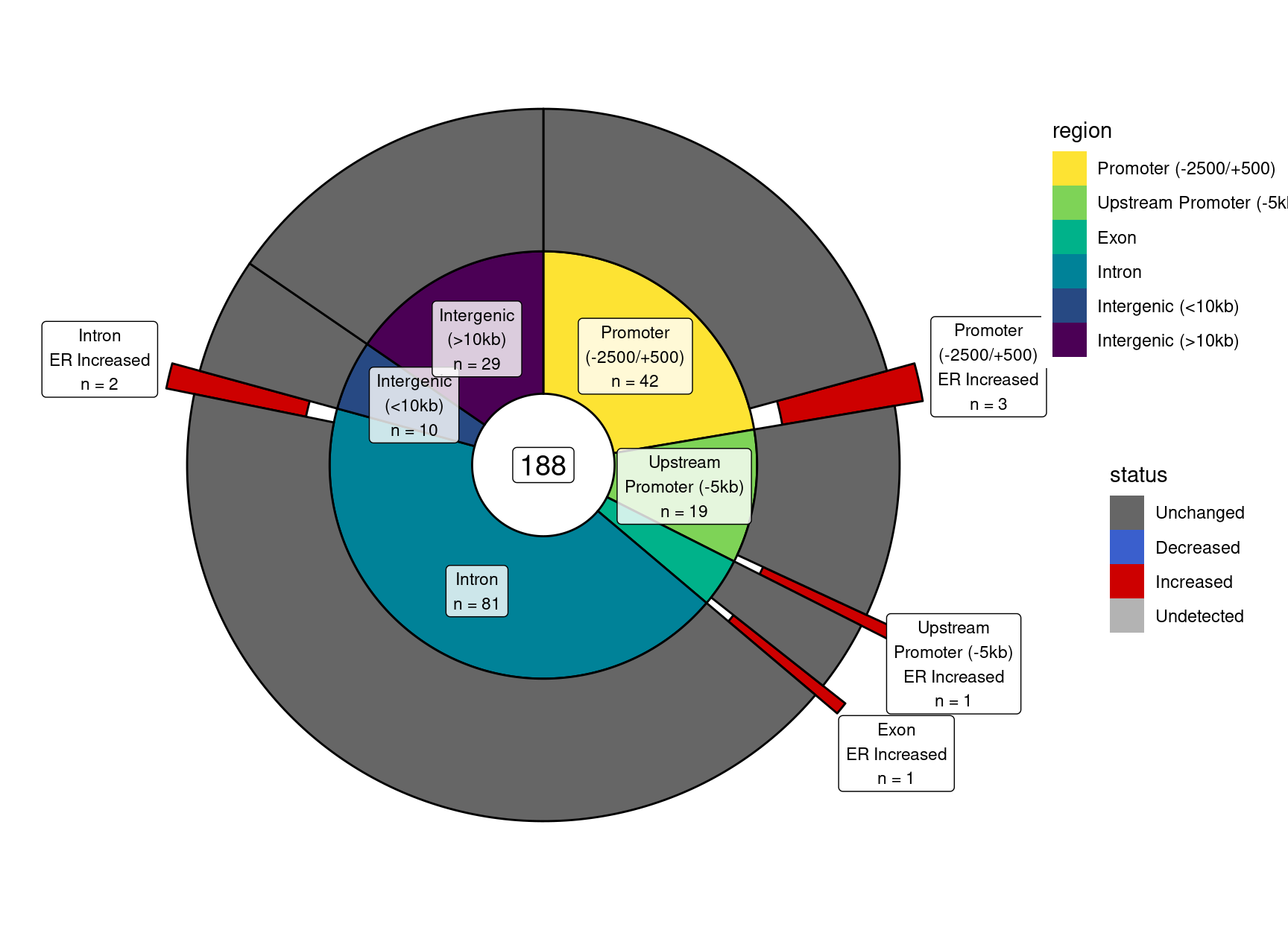
Chen, Yunshun, Aaron A T Lun, and Gordon K Smyth. 2016.
“From Reads to Genes to Pathways: Differential Expression Analysis of RNA-Seq Experiments Using Rsubread and the edgeR Quasi-Likelihood Pipeline.” F1000Research 5: 1438.
https://doi.org/10.12688/f1000research.8987.2.
Hahne, Florian, and Robert Ivanek. 2016.
“Statistical Genomics: Methods and Protocols.” In, edited by Ewy Mathé and Sean Davis, 335–51. New York, NY: Springer New York.
https://doi.org/10.1007/978-1-4939-3578-9_16.
Hickey, Theresa E, Luke A Selth, Kee Ming Chia, Geraldine Laven-Law, Heloisa H Milioli, Daniel Roden, Shalini Jindal, et al. 2021. “The Androgen Receptor Is a Tumor Suppressor in Estrogen Receptor-Positive Breast Cancer.” Nat. Med. 27 (2): 310–20.
Law, Charity W, Yunshun Chen, Wei Shi, and Gordon K Smyth. 2014. “Voom: Precision Weights Unlock Linear Model Analysis Tools for RNA-seq Read Counts.” Genome Biol. 15 (2): R29.
Lee, Stuart, Cook, Dianne, Lawrence, and Michael. 2019.
“Plyranges: A Grammar of Genomic Data Transformation.” Genome Biol. 20 (1): 4.
http://dx.doi.org/10.1186/s13059-018-1597-8.
Lun, Aaron T L, and Gordon K Smyth. 2016. “Csaw: A Bioconductor Package for Differential Binding Analysis of ChIP-Seq Data Using Sliding Windows.” Nucleic Acids Res. 44 (5): e45.
McCarthy, Davis J, and Gordon K Smyth. 2009. “Testing Significance Relative to a Fold-Change Threshold Is a TREAT.” Bioinformatics 25 (6): 765–71.
Ross-Innes, Caryn S., Rory Stark, Andrew E. Teschendorff, Kelly A. Holmes, H. Raza Ali, Mark J. Dunning, Gordon D. Brown, et al. 2012.
“Differential Oestrogen Receptor Binding Is Associated with Clinical Outcome in Breast Cancer.” Nature 481: –4.
http://www.nature.com/nature/journal/v481/n7381/full/nature10730.html.
Wickham, Hadley, Mara Averick, Jennifer Bryan, Winston Chang, Lucy D’Agostino McGowan, Romain François, Garrett Grolemund, et al. 2019.
“Welcome to the tidyverse.” Journal of Open Source Software 4 (43): 1686.
https://doi.org/10.21105/joss.01686.
Wilson, Daniel J. 2019. “The Harmonic Mean p-Value for Combining Dependent Tests.” Proc. Natl. Acad. Sci. U. S. A. 116 (4): 1195–1200.